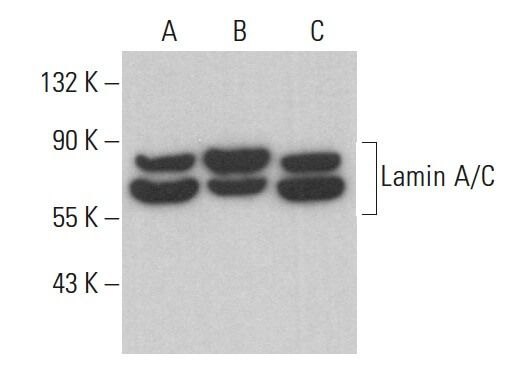
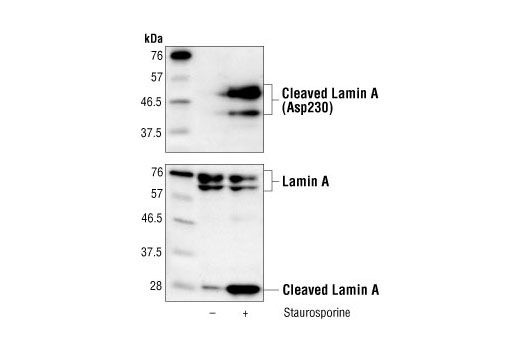
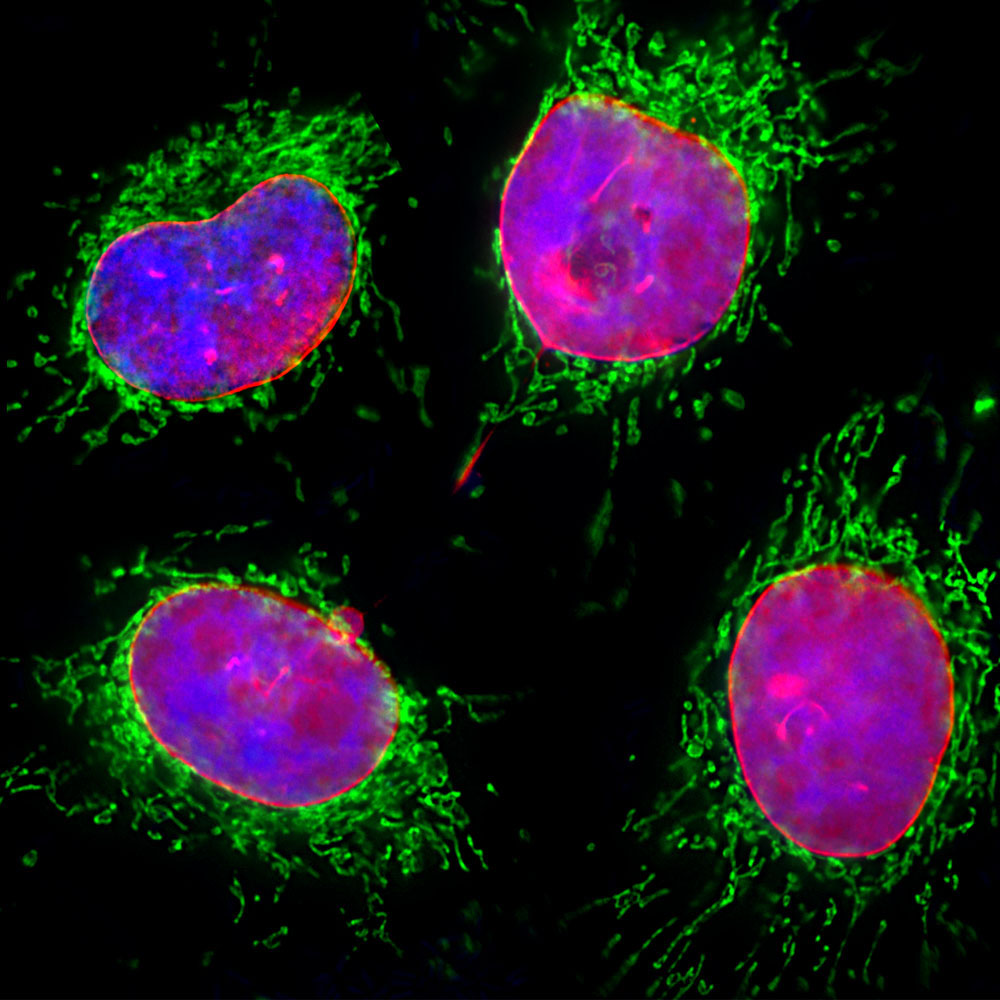
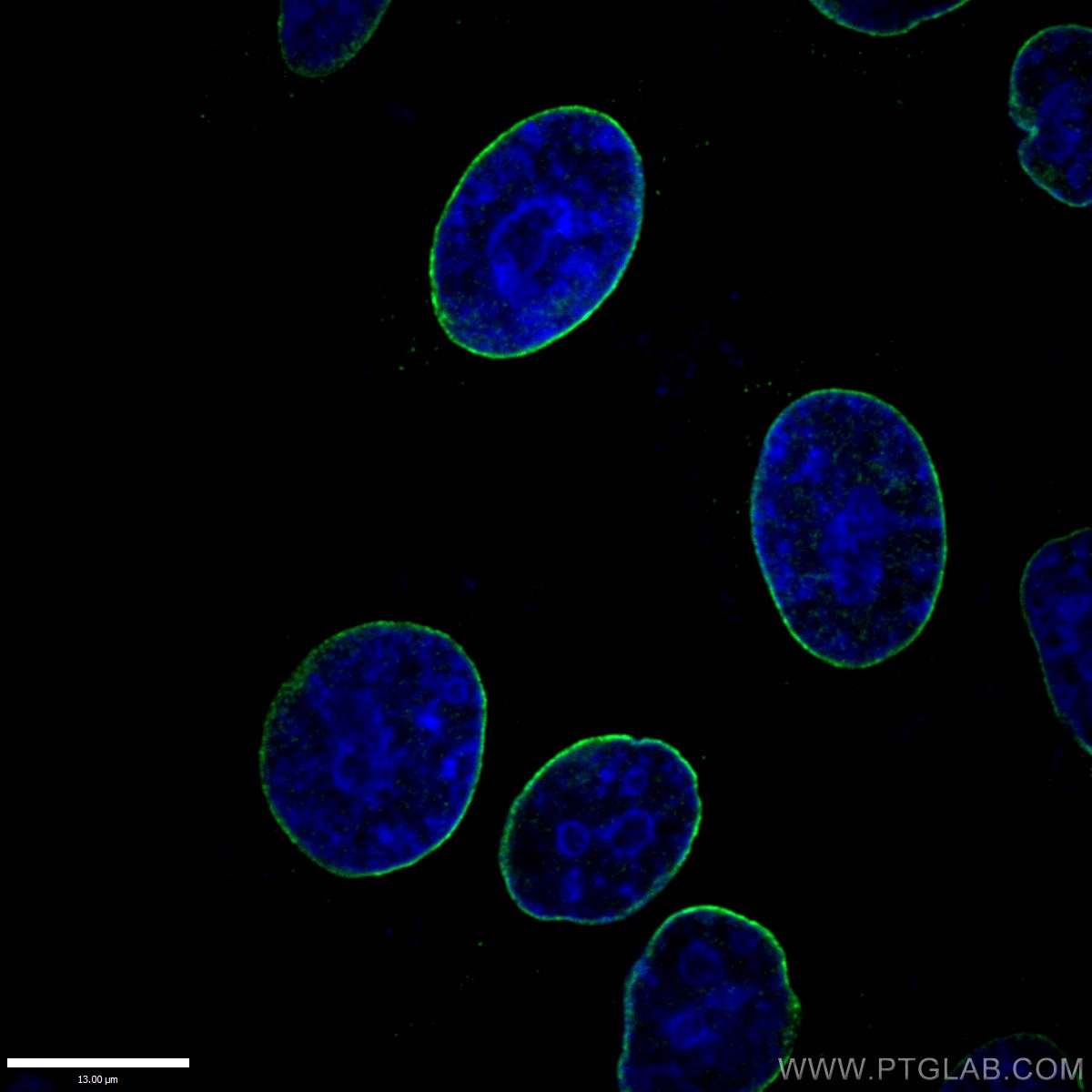
Missense Mutations in the Rod Domain of the Lamin A/C Gene as Causes of Dilated Cardiomyopathy and Conduction-System Disease | NEJM
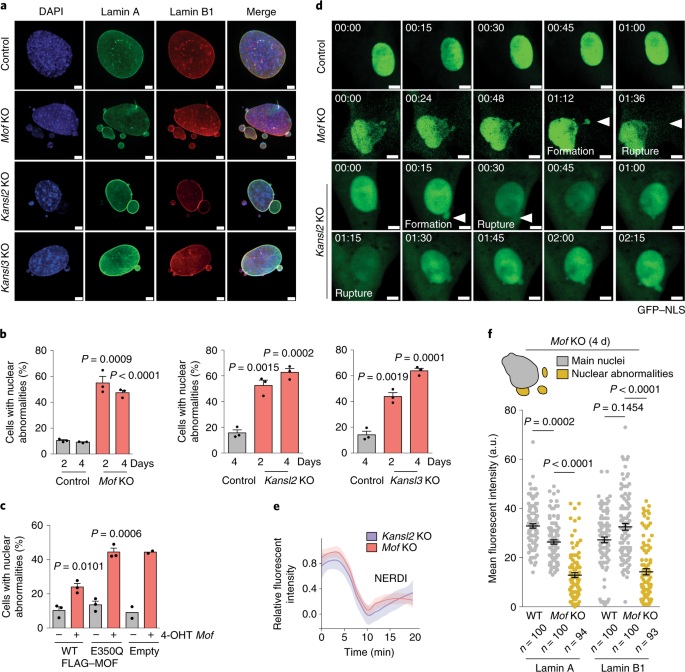
The NSL complex maintains nuclear architecture stability via lamin A/C acetylation | Nature Cell Biology
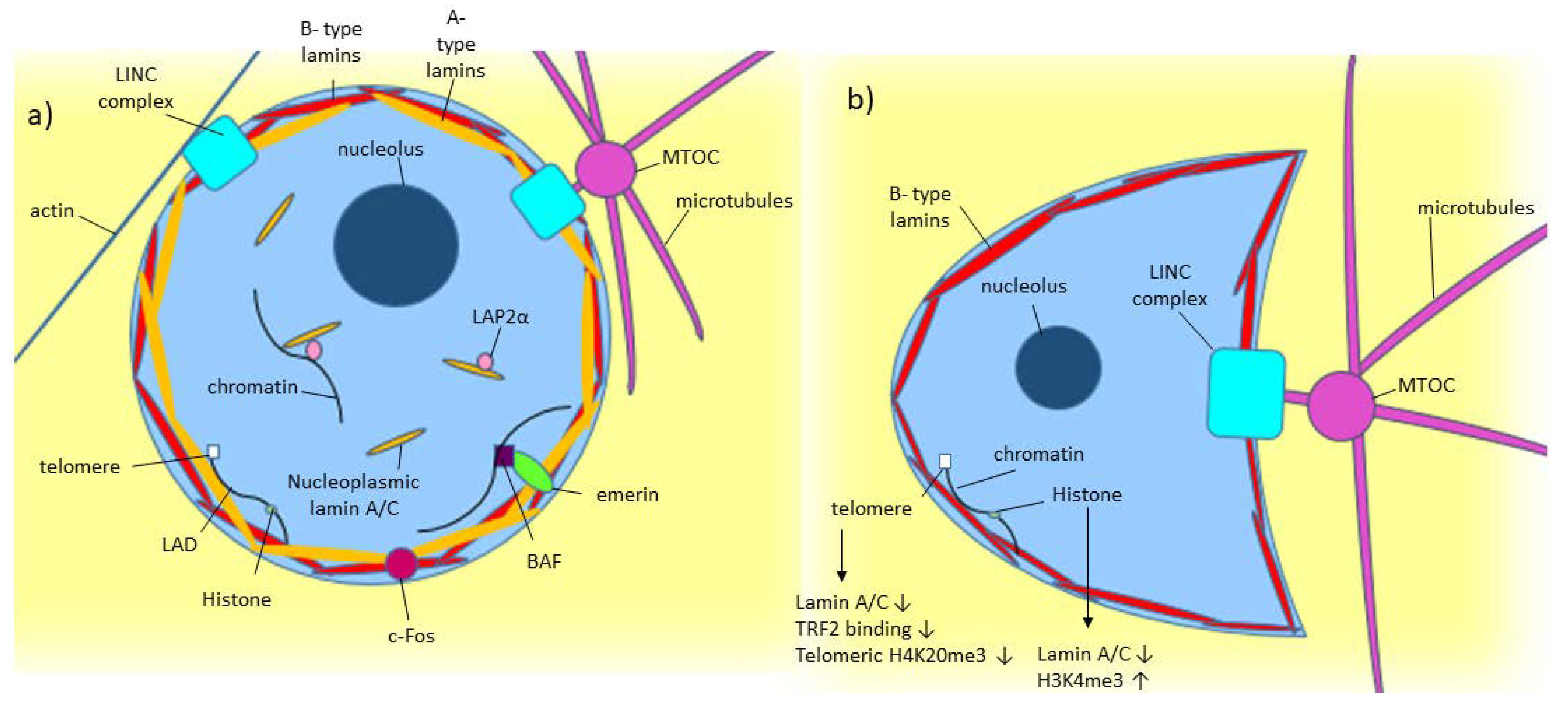
Frontiers | Diversity of Nuclear Lamin A/C Action as a Key to Tissue-Specific Regulation of Cellular Identity in Health and Disease
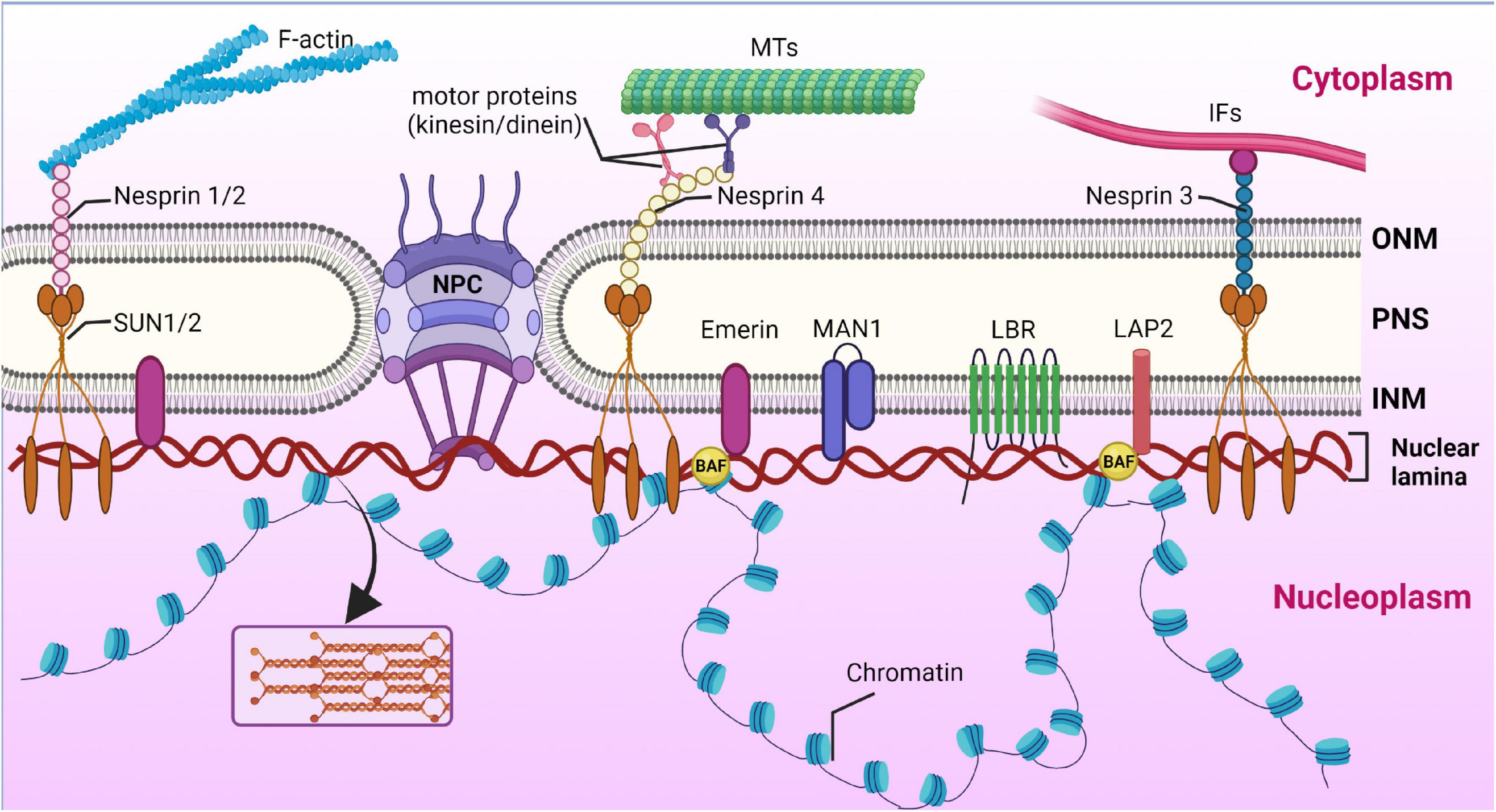
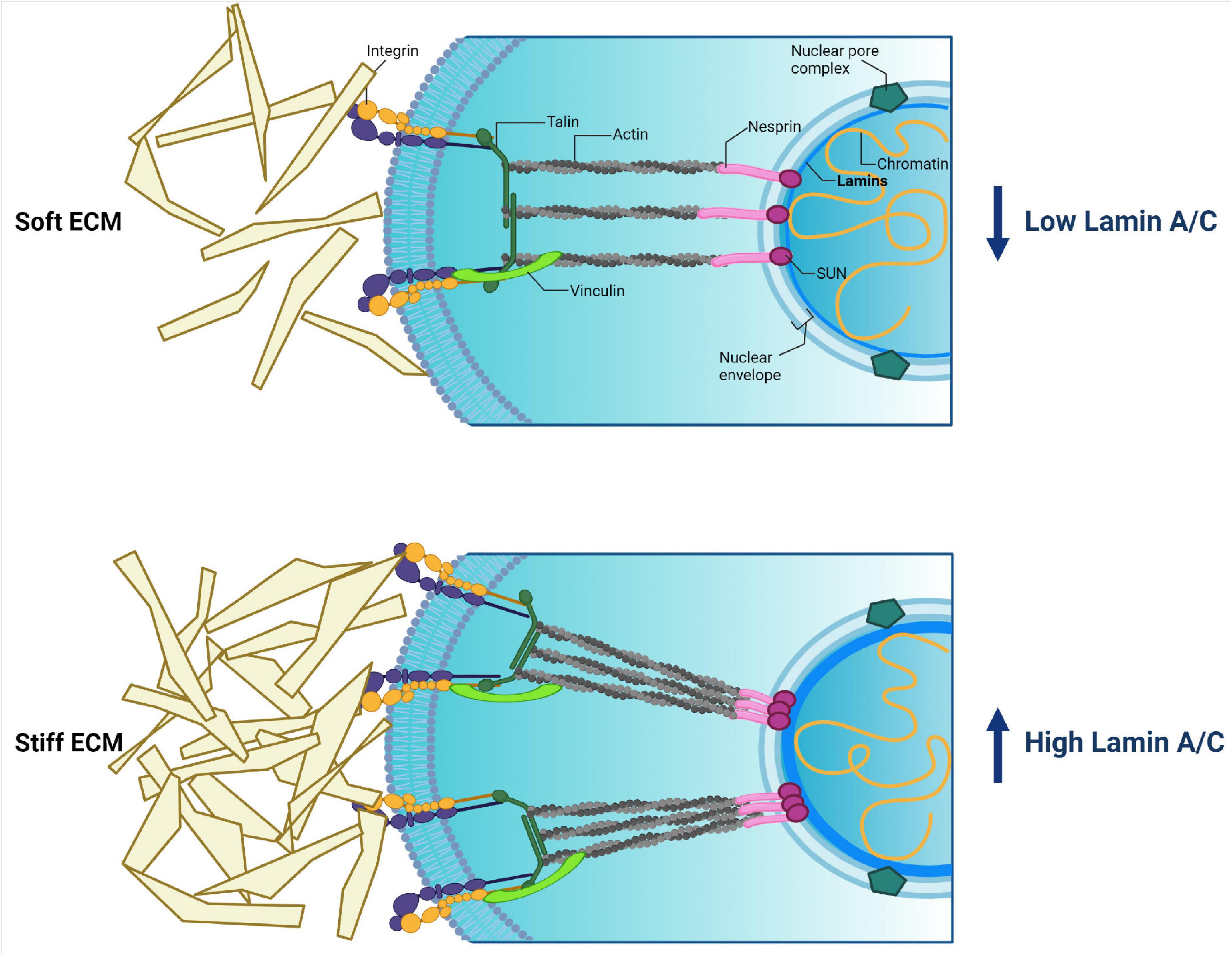
Frontiers | Diversity of Nuclear Lamin A/C Action as a Key to Tissue-Specific Regulation of Cellular Identity in Health and Disease

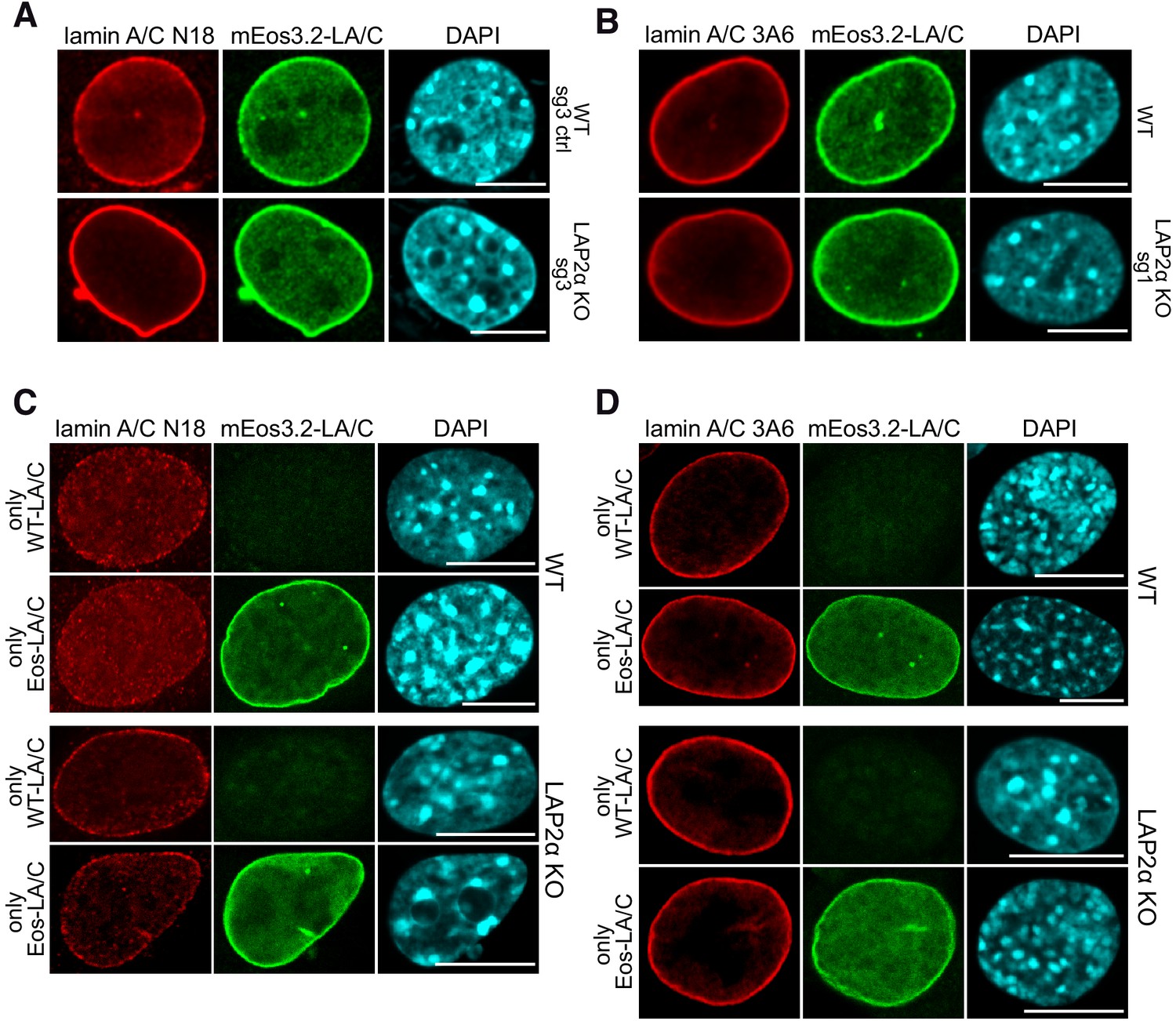
Phosphorylated Lamin A/C in the nuclear interior binds active enhancers associated with abnormal transcription in progeria | bioRxiv
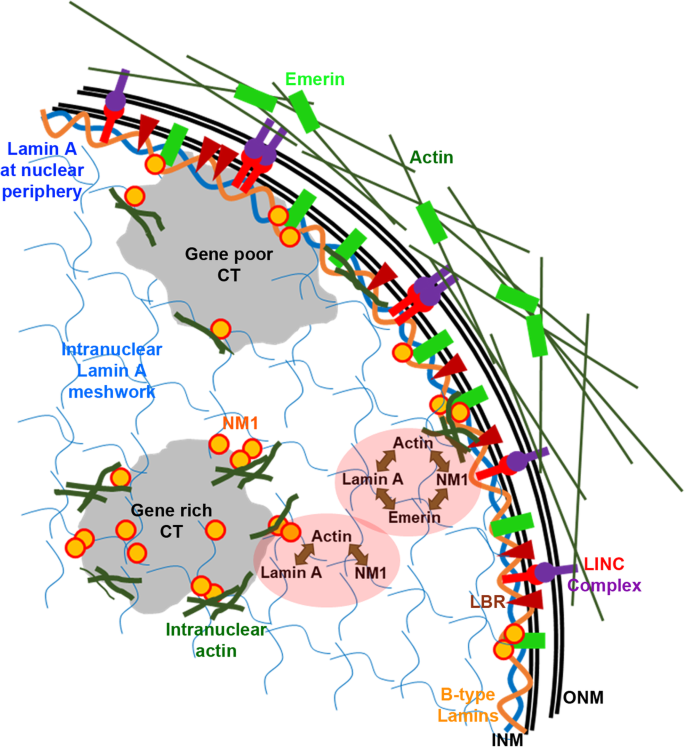
Lamin A/C and Emerin depletion impacts chromatin organization and dynamics in the interphase nucleus | BMC Molecular and Cell Biology | Full Text

Phosphorylated Lamin A/C in the Nuclear Interior Binds Active Enhancers Associated with Abnormal Transcription in Progeria - ScienceDirect